Pathway ID™ is designed on Model-Based Systems Engineering (MBSE) principles, providing a structured, compliant, and scalable approach to drug-device combinations that unlock new therapeutic potential.
Pathway ID™ is the entrypoint into DTx Discovery.
We integrate Pathway ID™ processes within Target Product Profiles (TPP) to ensure seamless compatibility and scalability for drug-device combinations. This approach allows for efficient development and ensures that the digital components align perfectly with drug TPPs, enabling cutting-edge, compliant, and adaptable solutions.
Get in touch to schedule a tailored primer workshop for your team. Designed for Business Development professionals, we’ll translate the science and risks behind DTx and smart medication into strategic pathways. Discover new possibilities.
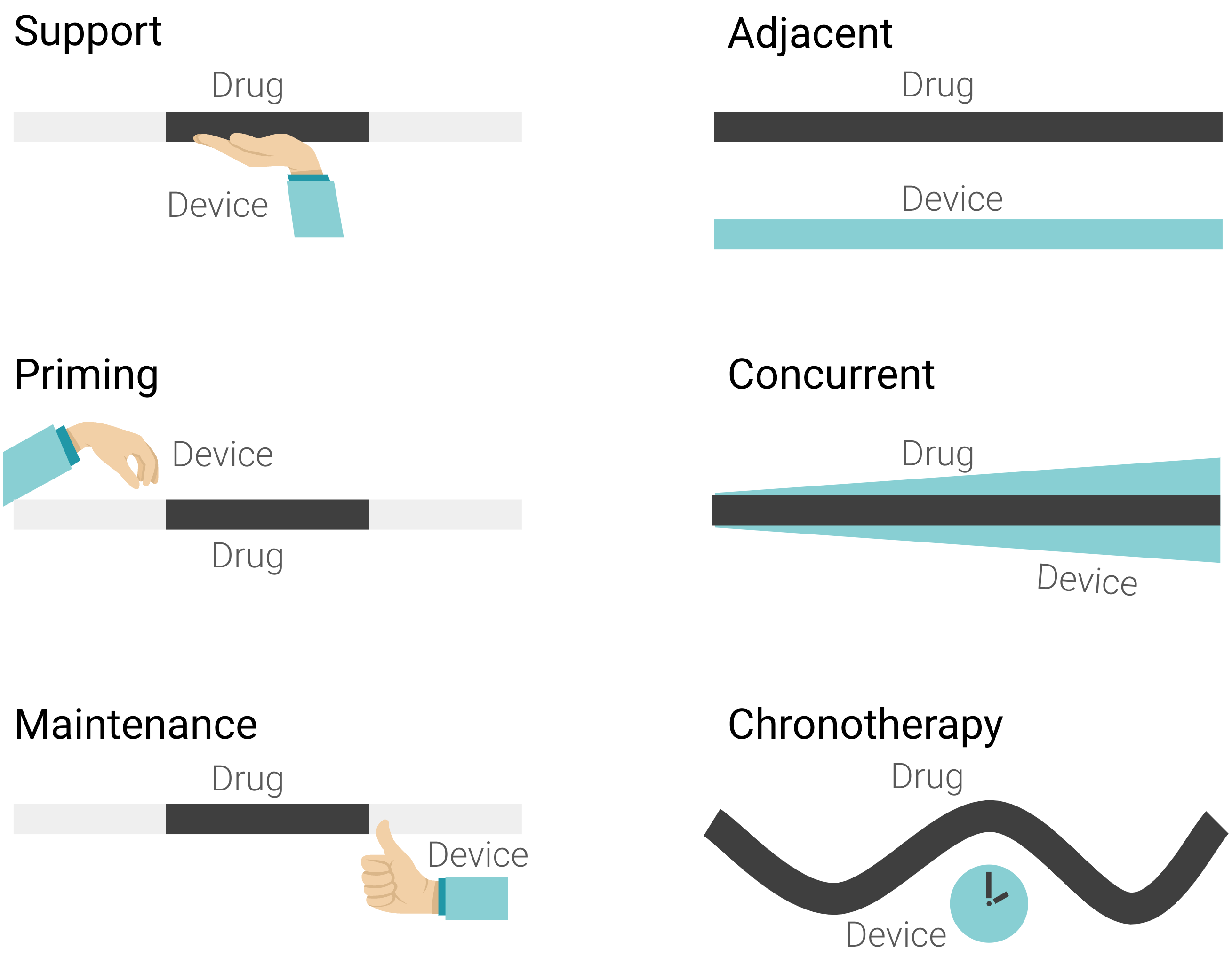
| Pathway ID™ Tier 1 | Pathway ID™ Tier 2 |
|---|---|
| Support | Adjacent |
| Priming | Concurrent |
| Maintenance | Chronotherapy |
| (Medical device risk class I & IIa) | (Medical device risk class IIb & IIb) |
By integrating devices with therapies, we drive innovation, enhance patient outcomes, and create long-term value. Our modular approach allows for seamless adaptation, ensuring that your products meet regulatory standards while remaining flexible for future advancements.
![]()
Smart Medications > Deep Dive > Systems Medicine
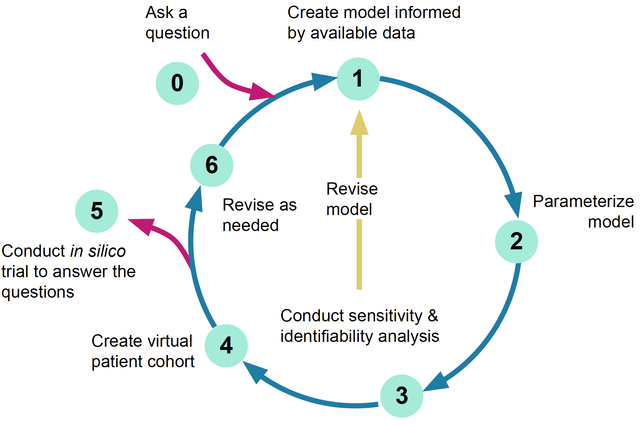 Virtual Population
Inhaltsangabe Migränegehirn
Lange galt Migräne als unsichtbare Krankheit. Nichtbetroffene unterstellten gerne auch schon mal, Migräne sei eine eingebildete Krankheit, denn es
Virtual Population
Inhaltsangabe Migränegehirn
Lange galt Migräne als unsichtbare Krankheit. Nichtbetroffene unterstellten gerne auch schon mal, Migräne sei eine eingebildete Krankheit, denn es